Changing Course
In my last post, I talked about how I was essentially replicating the Paloma article (previously linked) in a DIY manner. This is no longer the case.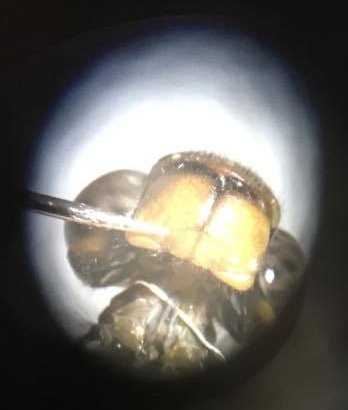
The primary goal at this point is to record from the TSDN tracts, located on the ventral portion of the dragonfly’s “neck” area (seen as two white lines in the photo), in a way that the presentation of a stimulus will elicit a selective response (hence the name target-selective descending neurons); that is, waving a dot left to right would get a response, but right to left would not. I have actually achieved this with a few dragonflies already, but right now instead of analyzing the data, I am working on improving my prep so that the dragonflies can be released more easily after recording.
As for the stimulus, I have been using flash cards with various sizes of dots to imitate a “fly”, as well as gradients. The laser rig idea has been scratched, and the newly revived suggestion of an iPad program is being considered. That will be tested soon and hopefully by the next post we have a working prototype of the stimulus that we end up with. Right now though, the hand-drawn dot works well enough to hear those pops from the spikerbox.

Directional Discrimination… White Event Markers are Left to Right stimulation… Orange, Right to Left
Hard-Won Wisdom
I have definitely improved my catching and prep techniques. First of all, the dragonflies in the arboretum tend to be in tall, grassy fields rather than along the river. There are hundreds upon hundreds of damselflies along the water, but rarely a dragonfly. Since catching them is very time consuming, we put out a local bounty for dragonflies. This past weekend, someone brought me in 11 dragonflies that they had caught. It was a busy weekend after that point.
Second, anesthetizing them in a refrigerator is good for storage, but not nearly long enough to keep them still during recording. I usually have to put them under ice water for about five minutes if they start moving around. As you can see in the earlier image above, the dragonfly is stuck to a popsicle stick with epoxy and further held down by silly putty. This setup is going to change again so that the dragonfly can be released more easily afterward – the (careful) removal of the glue doesn’t destroy any tissue, but residues might make it harder for them to fly. Also, during the prep, I shine a bright light on the dragonfly. This is essential. Without the bright light, there isn’t enough light reflecting off of the paper stimulus for the dragonfly to see it (cons of working in a basement), so there isn’t a response. The popsicle stick lies on half of a petri dish in order to elevate the dragonfly. The popsicle stick hangs off of the edge, and the dragonfly’s head hangs off of the edge of the popsicle stick, all so that the dorsal (back) side of its eyes have a clear view of the stimuli presented to it. I am still testing to see if it also needs to hang off of the desk to give it full vision below it (it is upside down, so “below” refers to the dorsal side). In the next week I hope to have perfected my methodology so I can start analyzing my data and begin writing up the results.
Sometimes Research is Fun!
I have done some pretty cool things with Backyard Brains these past few weeks. We went to see a Tigers game in the city, which was great fun, but they have nothing on the energy at Wrigley Field… For the Fourth of July, we took part in a parade to spread awareness about the company. I made huge dragonfly wings out of paper mache, and spent more time than I would have liked to painting and detailing them… I had a lot of fun talking to people about the company and getting the crowd enthusiastic about neuroscience. Some of the most meaningful experiences I’ve had working here has been talking to others about research, whether that was demoing my experiment at Tech Trek, getting stopped by arboretum visitors who are curious about my catches, or unexpectedly meeting people who use BYB products. I am really impressed with the connections we build as a result of our outreach.