-
 EducationWelcome to the cockroach world! My work for these five weeks was to develop a novel low-cost system to study the circadian rhythm of cockroaches. I’m working with my beautiful friends discoid cockroaches (B. discoidalis). Circadian rhythms are physical, mental, and behavioral changes that follow a roughly 24-hour cycle, responding primarily to light and darkness in an […]
EducationWelcome to the cockroach world! My work for these five weeks was to develop a novel low-cost system to study the circadian rhythm of cockroaches. I’m working with my beautiful friends discoid cockroaches (B. discoidalis). Circadian rhythms are physical, mental, and behavioral changes that follow a roughly 24-hour cycle, responding primarily to light and darkness in an […] -
 InternshipSo you’re trying to pick up Crickets? In this day and age there are services for everything; online dating for farmers, pastors, and anyone who’s looking for that special someone. Just nothing out there to help you find that very special cricket love. Well don’t worry, you won’t need a special site or even special skills! […]
InternshipSo you’re trying to pick up Crickets? In this day and age there are services for everything; online dating for farmers, pastors, and anyone who’s looking for that special someone. Just nothing out there to help you find that very special cricket love. Well don’t worry, you won’t need a special site or even special skills! […] -
 ExperimentHello again! This is the mind-reader reporting to you with updates on my project. I have had quite the scientific adventure since last sharing my research so sit down, grab your tea (or coffee or pop or kool-aide – I don’t judge) and prepare for a rollercoaster. With no success from LED oddball tasks, I moved […]
ExperimentHello again! This is the mind-reader reporting to you with updates on my project. I have had quite the scientific adventure since last sharing my research so sit down, grab your tea (or coffee or pop or kool-aide – I don’t judge) and prepare for a rollercoaster. With no success from LED oddball tasks, I moved […] -
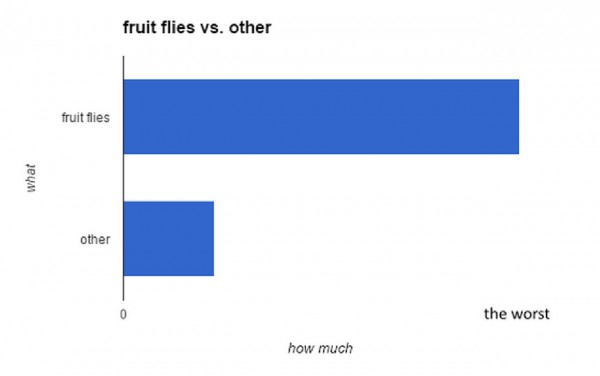 InternshipYou know what’s great about fruit flies? Nothing. fig. 1 Fruit flies suck Nothing, that is, other than their benefit as a model organism for simple and fast transgenic experimentation — but who really cares about all that. Drosophila melanogaster are butts, so what if they could die? Well they can (vinegar and plastic-wrap), we don’t need […]
InternshipYou know what’s great about fruit flies? Nothing. fig. 1 Fruit flies suck Nothing, that is, other than their benefit as a model organism for simple and fast transgenic experimentation — but who really cares about all that. Drosophila melanogaster are butts, so what if they could die? Well they can (vinegar and plastic-wrap), we don’t need […] -
 InternshipGreetings, this is Trevor coming live from Ann Arbor in a basement… We have one week left in the internship and things are finally starting to come together. Last time I made a post, I was without a doubt on the struggle bus in terms of getting data worthy of a poster, let alone a journal […]
InternshipGreetings, this is Trevor coming live from Ann Arbor in a basement… We have one week left in the internship and things are finally starting to come together. Last time I made a post, I was without a doubt on the struggle bus in terms of getting data worthy of a poster, let alone a journal […] -
 InternshipHello my name is Bailey! I am a junior majoring in electrical engineering at Michigan State University and am doing an internship at Backyard Brains this summer. Sorry I missed the first blog post, I was travelling in Japan with my sister! I know it doesn’t look like it from the picture, but I was doing […]
InternshipHello my name is Bailey! I am a junior majoring in electrical engineering at Michigan State University and am doing an internship at Backyard Brains this summer. Sorry I missed the first blog post, I was travelling in Japan with my sister! I know it doesn’t look like it from the picture, but I was doing […] -
 HardwareName: Katelyn Rowley School: University of Michigan (Go Blue!) Major: Biomedical Engineering Hobbies: Running (I’ve run a half marathon and I hope to run a full one someday!), journaling, trying new restaurants in Ann Arbor, being outdoors, finding new music to listen to (Florence and the Machines, Bon Iver, classic rock, you name it) What’s up interwebz? When I […]
HardwareName: Katelyn Rowley School: University of Michigan (Go Blue!) Major: Biomedical Engineering Hobbies: Running (I’ve run a half marathon and I hope to run a full one someday!), journaling, trying new restaurants in Ann Arbor, being outdoors, finding new music to listen to (Florence and the Machines, Bon Iver, classic rock, you name it) What’s up interwebz? When I […] -
 InternshipHey! What’s up? My name is Trevor Smith, currently a senior at the fabulous Michigan State University, and I am lucky enough to be participating in an internship at Backyard Brains this summer. I am currently working on pheromone detection in moth antennae, specifically how sensitive male moths antennae are to the female pheromone used […]
InternshipHey! What’s up? My name is Trevor Smith, currently a senior at the fabulous Michigan State University, and I am lucky enough to be participating in an internship at Backyard Brains this summer. I am currently working on pheromone detection in moth antennae, specifically how sensitive male moths antennae are to the female pheromone used […] -
 InternshipMy name is Nick Weston and I am an intern in the summer program at Backyard Brains. I’m an an undergraduate student studying neuroscience at Michigan State University and during this internship I plan on trying to capture neuronal spiking activity from the internal organs of a crickets ear while also trying to record and […]
InternshipMy name is Nick Weston and I am an intern in the summer program at Backyard Brains. I’m an an undergraduate student studying neuroscience at Michigan State University and during this internship I plan on trying to capture neuronal spiking activity from the internal organs of a crickets ear while also trying to record and […] -
 EducationWelcome! This is Kylie Smith, a Michigan State University undergraduate writing to you from a basement in Ann Arbor. I am studying behavioral neuroscience and cognition at MSU and have been fortunate enough to have landed an internship with the one and only Backyard Brains for the summer. I am working on The Consciousness Detector […]
EducationWelcome! This is Kylie Smith, a Michigan State University undergraduate writing to you from a basement in Ann Arbor. I am studying behavioral neuroscience and cognition at MSU and have been fortunate enough to have landed an internship with the one and only Backyard Brains for the summer. I am working on The Consciousness Detector […] -
 EducationComing soon to a backyard near you. At least, that’s the idea. We’re sure the technology will catch up if we give it enough prodding and throw an intern or two its way. And hey if not? There’s still lasers, sounds like a win/win to me. Wait we don’t get lasers either? This is really going […]
EducationComing soon to a backyard near you. At least, that’s the idea. We’re sure the technology will catch up if we give it enough prodding and throw an intern or two its way. And hey if not? There’s still lasers, sounds like a win/win to me. Wait we don’t get lasers either? This is really going […]