-
 FellowshipWhat follows is a tale of customs adventures, personal legal status in a foreign country, and the history of Serbia and Yugoslavia. We have had the grace to work in Serba a number of times over the past few years, and 2024 was no different. During July, we were invited by the Center for the […]
FellowshipWhat follows is a tale of customs adventures, personal legal status in a foreign country, and the history of Serbia and Yugoslavia. We have had the grace to work in Serba a number of times over the past few years, and 2024 was no different. During July, we were invited by the Center for the […] -
 EducationIn an article we previously published in June 2022 about our scientific paper that dealt with play behavior in fish, I concluded at the end of the article: “I think it is possible for novices and high school students to publish papers (and it is the dream and goal of our team)… That is why […]
EducationIn an article we previously published in June 2022 about our scientific paper that dealt with play behavior in fish, I concluded at the end of the article: “I think it is possible for novices and high school students to publish papers (and it is the dream and goal of our team)… That is why […] -
 EducationBackyard Brains is now in its second year of interns from the University of Santiago de Chile (affectionately called Usach). Last year we had a project recording the ganglia of snails – and this we will continue our voyage in the world of invertebrates with an old favorite and a new favorite. Cockroaches and Clams. […]
EducationBackyard Brains is now in its second year of interns from the University of Santiago de Chile (affectionately called Usach). Last year we had a project recording the ganglia of snails – and this we will continue our voyage in the world of invertebrates with an old favorite and a new favorite. Cockroaches and Clams. […] -
 EducationHi Everyone. Juan here! My two month tour with Backyard Brains has reached its end, and I’m really grateful to have had the opportunity to work on this project. I had three activites during the “practica” here at Backyard Brains: Recording from the Ganglia of Snails Helping on the Anemone Project Assisting in Outreach. The […]
EducationHi Everyone. Juan here! My two month tour with Backyard Brains has reached its end, and I’m really grateful to have had the opportunity to work on this project. I had three activites during the “practica” here at Backyard Brains: Recording from the Ganglia of Snails Helping on the Anemone Project Assisting in Outreach. The […] -
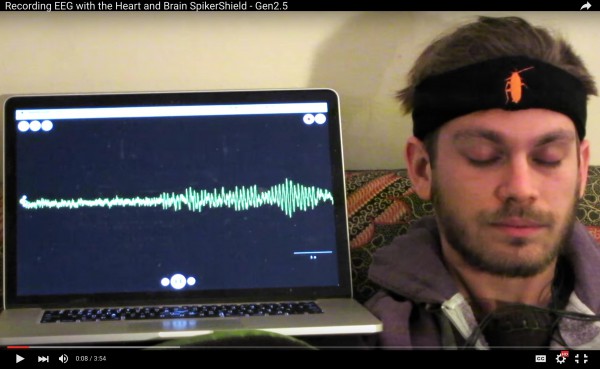 BizAs part of our operations, we give talks to the public on weekly basis. We begin with neural signals from the legs of cockroaches, display of which carries low risk as the legs can grow back and cockroach leg neural signals are not covered under United States Privacy laws. But, as we have been expanding […]
BizAs part of our operations, we give talks to the public on weekly basis. We begin with neural signals from the legs of cockroaches, display of which carries low risk as the legs can grow back and cockroach leg neural signals are not covered under United States Privacy laws. But, as we have been expanding […] -
 HardwareLast year, we launched our “human human interface” allowing a connection between complete strangers to be built artificially, bypassing chance. But there are those lucky people who don’t need a mechano-electrical connection, who say they simply “feel something” in the presence of another. Being in the same room together, subjects report feeling “warmer,” that there […]
HardwareLast year, we launched our “human human interface” allowing a connection between complete strangers to be built artificially, bypassing chance. But there are those lucky people who don’t need a mechano-electrical connection, who say they simply “feel something” in the presence of another. Being in the same room together, subjects report feeling “warmer,” that there […] -
 EducationOn January 1st, we received a New Year’s gift from another continent: Neuroscience tools and experiments made by a group of high school students selected from the 20 best rated schools of Iran. They were written lab reports, submitted for an interdisciplinary neuroscience competition that utilized our open source experiments with cockroaches as a resource […]
EducationOn January 1st, we received a New Year’s gift from another continent: Neuroscience tools and experiments made by a group of high school students selected from the 20 best rated schools of Iran. They were written lab reports, submitted for an interdisciplinary neuroscience competition that utilized our open source experiments with cockroaches as a resource […] -
 EducationThis past August, we were graciously invited by ZAST (Zhejiang Association for Science and Technology) to come to China to give a series of workshops and talks in Hangzhou, Ningbo, and Shanghai at the various impressive science museums there. Similarly to the United States, Neural Engineering is a relatively new discipline for the public, and […]
EducationThis past August, we were graciously invited by ZAST (Zhejiang Association for Science and Technology) to come to China to give a series of workshops and talks in Hangzhou, Ningbo, and Shanghai at the various impressive science museums there. Similarly to the United States, Neural Engineering is a relatively new discipline for the public, and […] -
 Education**Edit: This design is no longer available as of 2020** We at Backyard Brains love cockroaches. Every time we see someone with butterfly earrings or a butterfly tattoo, we think, “That would look so much better if it were a cockroach. Pobrecita Cucaracha. Nobody celebrates you.” No more, you can now show your love of […]
Education**Edit: This design is no longer available as of 2020** We at Backyard Brains love cockroaches. Every time we see someone with butterfly earrings or a butterfly tattoo, we think, “That would look so much better if it were a cockroach. Pobrecita Cucaracha. Nobody celebrates you.” No more, you can now show your love of […] -
 EducationIn March 2014 we went to Mexico’s first Maker Faire, invited by Amor Muñoz, a Mexican Textile and Electronic artist, who we met at Chile’s Santiago MakerFaire in November 2013. The Mexico MakerFaire took place in Oaxaca, a place known for traditional crafts, pero ahora se esta transformando en un lugar de “Nuevos-Makers” de diseño, […]
EducationIn March 2014 we went to Mexico’s first Maker Faire, invited by Amor Muñoz, a Mexican Textile and Electronic artist, who we met at Chile’s Santiago MakerFaire in November 2013. The Mexico MakerFaire took place in Oaxaca, a place known for traditional crafts, pero ahora se esta transformando en un lugar de “Nuevos-Makers” de diseño, […] -
UncategorizedWe are in Boston at NSTA (National Science Teacher’s Association), booth 1432, from Thursday 11 AM – Saturday 5 PM. We also have workshops Saturday in room 107A at 8 AM and 2 PM. Come by, say hello, and see some new SpikerShield and Microscope Inventions.
-
 UncategorizedOf the many problems that exist in this world, many would agree that at a personal level… the most drastic problem is the lack of connection between people. The probability of a marriage ending in divorce is 40-50%, with many divorcees claiming that “the feeling simply went away,” disappearing into the ether of space. The […]
UncategorizedOf the many problems that exist in this world, many would agree that at a personal level… the most drastic problem is the lack of connection between people. The probability of a marriage ending in divorce is 40-50%, with many divorcees claiming that “the feeling simply went away,” disappearing into the ether of space. The […]